![]()
![]()
Questions? Call Us 1–773–271–0318 9am to 2pm Central Time Mon.–Fri.
The Better Classroom Introduction of the Periodic Table
The value of beginning learning about chemistry with a 3D periodic table can't be overestimated.
The familiar printed icon of chemistry can not be readily replaced by a dimensional object, but, during introduction to chemistry, will help make sense of the need for breaks and gaps in the familiar form, flattened for convenience of printing and distribution – much as printed world geographic projections (maps) are far handier than globes for general use.

The very first accepted periodic table was based on the premise that the elements should naturally be in unbroken order, but still allow for the (periodically) related properties be aligned as well - which just turned out to need all three dimensions. Alexandre–Emile Beguyer de Chancourtois did this in 1862, wrapping the element indicators on the outside of a tube, in a helix.
Since then, for convenience of printing of books and posters, the flat chart has become so ubiquitous that most assume that this is the proper representation. That would not be so bad, except that this flawed image (propagated by the International Union of Pure and Applied Chemistry, the accepted authority) is both confusing and disjointed, and when one begins to relate it with the Periodic Law, the table is also simply wrong in its many departures from the Law. By providing an unstable basis for future learning, and introducing (reinforcing?) doubt in authority, the credibility of the flat table is challenged.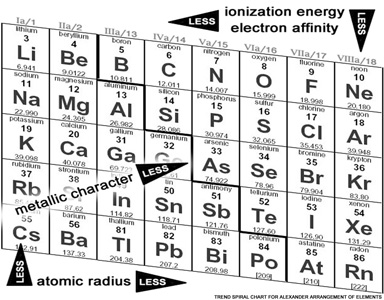
All the varied static and/or interactive flat periodic table charts – for myriad reasons – must be used and relied upon if anyone is to study chemistry seriously. The 3D periodic table is best applied in the initial introduction to the periodic table to new students, as the world globe initiates the understanding of distortions ('projections') required in printing world maps – 3D is the reality, flat printed adaptations are for convenience, economy, and work.
We have developed lesson plans and teacher support material - plus some PowerPoints - to help your students to learn about the periodic table easier, faster, and better, in accompaniment with the Alexander Arrangement of Elements in the classroom.
Teaching Ideas for Using the Alexander Arrangement of Elements in Class
< previous
© 2014 AlexanderDESIGN
4851 N. Washtenaw Ave., Chicago, IL 60625 773.271.0318