The Three–Dimensional Periodic Table
The Educational Concept of the Alexander Arrangement of Elements 3D Periodic Table

An examination of the accuracy and educational value
of a three–dimensional periodic table of the elements.
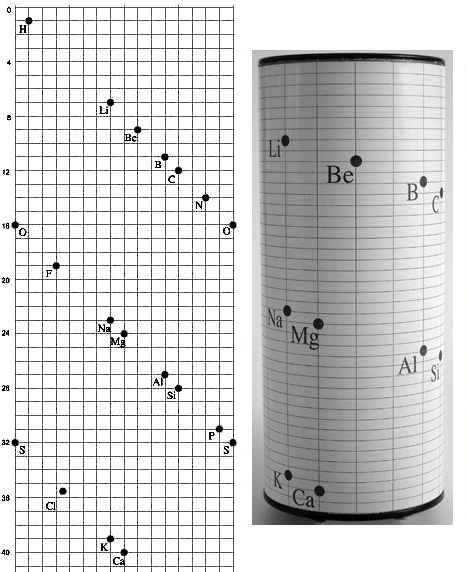
The periodic table model in the accompanying photos will, I expect, if used in the classroom for the lesson prior to that of introduction to the flat periodic table, promote quick and easy familiarity with, and credibility for, the periodic table in its standard flat form.
Strict and literal conforming to the Periodic Law's primary tenet "that all elements be arranged in order" was the inspiration and primary factor for its development by myself, Roy Alexander.
The secondary feature of this model is to clearly and physically define the electron configuration blocks of the periodic table.
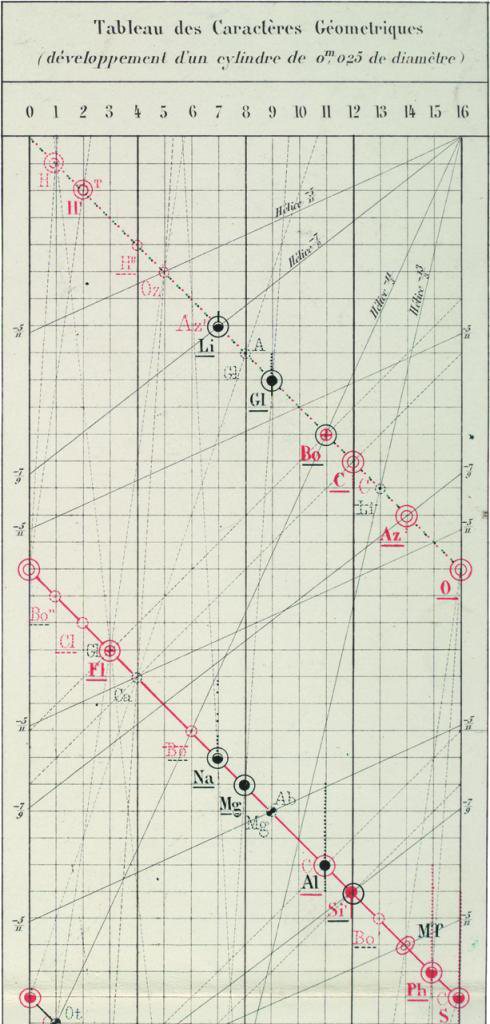
Thirdly, I have discovered, is historic; its form is directly related that of the first periodic table – seven years before Mendeleev's – the three–dimensional vis Tellurique of Alexandre–Emile Beguyer de Chancourtois.
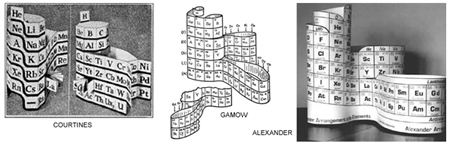
Research has also revealed to me several other apparently independently arrived at periodic table versions
developed in the 20th century similar to mine; Janet's, Courtine's, Gamov's, Denker's, Olga's, Giguere's and possibly others some earlier, some later. The Alexander Arrangement was patented.
Rotation of the 3D table pictured below permits a complete view of all the data, promoting hands–on interaction by the student – adding tactile and kinetic experiences to the intellectual exercise just of looking at it, not to mention the tactile and kinetic experiences of assembling it. The novel and interesting look of the table is a major attraction for many non–academically inclined students.
I am attempting to promote the use of this 3D periodic table in schools just before the periodic table is introduced. As a respected expert regarding the periodic table, I hope that you can find a few minutes to identify for me any statements I make here regarding my 3D table that are faulty, and inform me.
(The following images after #1 are of the Alexander Arrangement of Elements, each rotated clockwise 45� from the previous image.)
Image #1
The fold at the left of the s – block marks the beginning and end of the periods where they are joined with atomic numbers contiguous, a continuity consistent throughout this 3D model.
A fold at the right end of the top rows of the s – block is the other joining with the p – block.
The f– and d – blocks each loop out from the intersection of groups 2 and 3 in this Y over Lu Alexander Arrangement. The normally detached f – block is now returned to its proper place with elements at both ends adjacent to their neighbors in sequence.
Image #2
The s – block includes H with the noble gases. The start of an extension to the H data box can be seen. This will lend H to the p – block (see image #8).
The teardrop shape of the f – block is clear, as is the perpendicular connection with the remainder of the table for best viewing of the element data, aided by the pinch of the element boxes closest to the joining.
Image #3
The element data visibility mentioned above is seen here, and the joining of the s– and – block becomes evident.
The s– and d – blocks are in the same plane, their division indicated by the f – block as well as the vertical dimension.
Image #4
The separation of the H box extension is in full view here. In some product versions, the extension touches C in mid loop, as it addresses the contention that H has relations with C, He, Ne, F, Li, and Be, all of which are now, at minimum, corner connected to H.
The repair of the gap between groups 2 and 13 is clear.
The loop of the d – block is less generous, as the number of elements is fewer.
Image #5
Evident is the multi–helical aspect of the p – block, key to the uniqueness of this 3D model, as, reminiscent of the very earliest numerically based element arrangement, it also forms a helix.
The three physical blocks are portrayed well in this photo, and the s – block can be seen returning where it should, with the bend at groups??? 12 and 13 marking the block division.
Image #6
The S – block is hidden in this view, and most of the f – block as well.
The H box extension is well defined. In some versions it is looped above Li as well as over the p – block.
Image #7
The p – block dominates this image, and shows the distinct similarity with de Chancourtois 3D model, a rotatable tube in a with the element indications in a spiral or helix.
The f – block begins to show on the right.
Image #8
Here we see the end of the periods, much of the f – block, and the H box terminating completely at He – and peripherally contacting F and Ne.
AlexanderDESIGN 4851 N. Washtenaw Ave., Chicago, IL 60625 773.271.0318 last update 4/18/12