Alexandre–Emile
Beguyer
de Chancourtois
and the first
periodic table

An 1860 science conference produced a much more accurate list of atomic weights of the known elements than previously available.
Using this development, Alexandre de Chancourtois, of France, listed them in order of increasing weight of their atoms, the first person to do so.
And then, in 1862, a year before Newlands published his classification of the elements, and about 7 years prior to the announcement of Meyer’s and Mendeleev’s periodic tables, de Chancourtois created a fully functioning and unique system of organizing the chemical elements, inventing a spiral graph with the elements arranged on a cylinder for proper continuity of the spiral, which allowed placing the element series in unbroken order. He used his previous sequence – increasing atomic weight – and lined up similar elements vertically, developing a circumference of 16 units, 16 being approximately the atomic weight of oxygen.
De Chancourtois then stated that "the properties of the elements are the properties of numbers", and was first to recognize that elemental properties reoccur every seven elements – the basic principle of all succeeding periodic tables. Using this chart, he was able to predict the stoichiometry (the math behind chemistry) of several metallic oxides.
Despite this, his invention attracted little attention. The original scientific paper – presented to the French Academy of Sciences – was published without the diagram, making the revolutionary concept hard to comprehend. The diagram did, however, appear in a less widely read geological pamphlet, dealing mainly with geological concepts unfortunately, not suited to the interests of many chemists.
Mendeleev ...the most popular periodic table
Not until 1869 and later, when Dmitri Mendeleev's periodic table attracted attention and gained widespread scientific acceptance, was the value of de Chancourtois’ earlier periodic table recognized.
Latter day inventors
...better for teaching & learning
Several scientists, and a science exhibit designer revived the 3D periodic table concept in the 20th Century. Some have started with a ribbon of elements in atomic number sequence and wrapped it in a spiral to vertically align elements with similar properties, which establishes the ‘periodic’ nature of the table. Others may have merely wrapped the plane of the flat table (after ramping the element rows – escalator–like – in the p–block )
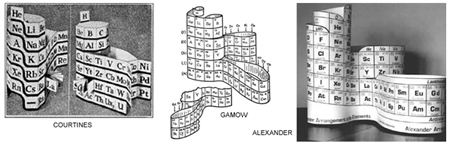
and let the post–Emile/Dmitri element blocks loop to make adjacent a perfect atomic number sequence. Some may have been solving technical questions, and others, like Mendeleev, Gamov and myself, trying to prepare a better educational tool.
Receiving a patent, I suppose, designates the applicant as an inventor.
I (Roy Alexander) created the 3D periodic table I call the Alexander Arrangement of Elements just because it was correct, and patented it much later.