
 I am primarily a museum science exhibit designer. In a design office where I worked in 1965 I saw a listing of a stack of drawings by another designer that identified one of the drawings as '3D Periodic Table'.
I am primarily a museum science exhibit designer. In a design office where I worked in 1965 I saw a listing of a stack of drawings by another designer that identified one of the drawings as '3D Periodic Table'.
I had recently been having discussions with friends concerning concepts of reality, and had concluded that all real things must have at least three dimensions, and was convinced that the periodic table, of all things, was real – what reflected reality more? ...or more of reality!
|
|
 I eagerly searched up the drawing, but was disappointed to find it to be a standard flat table with raised letters.
I eagerly searched up the drawing, but was disappointed to find it to be a standard flat table with raised letters.
 But I was already hooked on the 3D periodic table idea. I 'knew' the design concept was valid, and in an hour or so of
experimentation, proved it, placing elements in a continuous ribbon of databoxes by wrapping and looping, to make perfectly adjacent property groups. The table needed only one adjustment - years later - the downslant of element data boxes within the Main Group so each period descends to meet the start of the one below, maintaining the
helical form and eliminating the dozen+ breaks in the atomic number sequence of periodic tables.
But I was already hooked on the 3D periodic table idea. I 'knew' the design concept was valid, and in an hour or so of
experimentation, proved it, placing elements in a continuous ribbon of databoxes by wrapping and looping, to make perfectly adjacent property groups. The table needed only one adjustment - years later - the downslant of element data boxes within the Main Group so each period descends to meet the start of the one below, maintaining the
helical form and eliminating the dozen+ breaks in the atomic number sequence of periodic tables.
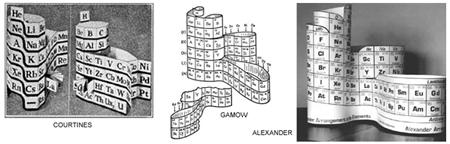
 At the time I had no idea that others (above: Courtines', Gamow's and mine) had developed a similar technique to build a 3D table – including Alexandre-Emile Beguyer de Chancourtois, who is credited with being the first to publish a periodic table showing the periodicity of element properties.
At the time I had no idea that others (above: Courtines', Gamow's and mine) had developed a similar technique to build a 3D table – including Alexandre-Emile Beguyer de Chancourtois, who is credited with being the first to publish a periodic table showing the periodicity of element properties.
 The Alexander Arrangement form brings the Seaborg created f–block into the table rather than being awkwardly orphaned. This, and the more modern way of thinking about Hydrogen – as being in more families than one – again affirms the advantages of three dimensions.
The Alexander Arrangement form brings the Seaborg created f–block into the table rather than being awkwardly orphaned. This, and the more modern way of thinking about Hydrogen – as being in more families than one – again affirms the advantages of three dimensions.
|
|
 Subsequent study of the Periodic Law and the periodic table's enormous value in education revealed that my basic rationale for developing the Alexander Arrangement of Elements and producing it was to support the Periodic Law rather than supplanting the PT, so I applied for and was granted a U.S. patent on the downslant in order to produce the AAE teaching/learning aids.
Subsequent study of the Periodic Law and the periodic table's enormous value in education revealed that my basic rationale for developing the Alexander Arrangement of Elements and producing it was to support the Periodic Law rather than supplanting the PT, so I applied for and was granted a U.S. patent on the downslant in order to produce the AAE teaching/learning aids.
 Initially, my goal was introduction of the AAE into classrooms, laboratories, chemistry textbooks, and reference material, universally replacing the conventional flat form.
Initially, my goal was introduction of the AAE into classrooms, laboratories, chemistry textbooks, and reference material, universally replacing the conventional flat form.
 After a decade of investigation of chemistry & science curricula, however,it has become clear that a 3D periodic table showing the element line intact , is best applied to prove the Periodic Law, prior to the introduction of the Periodic Table to new students, much as the world globe initiates the study of world maps – 3D is the reality, and flat printed adaptations are projections of that to paper for convenience, economy, and common use.
After a decade of investigation of chemistry & science curricula, however,it has become clear that a 3D periodic table showing the element line intact , is best applied to prove the Periodic Law, prior to the introduction of the Periodic Table to new students, much as the world globe initiates the study of world maps – 3D is the reality, and flat printed adaptations are projections of that to paper for convenience, economy, and common use.
 The familiar icon of chemistry, forced into awkwardness by the feature of showing "all the elements all the time", can not be readily replaced for learning and work, but the Alexander Arrangement, taught first, preserves the Table's validity, making sense of the need for breaks and gaps in the familiar form – flattened for convenience of printing and distribution – as printed projections of the World on maps are far handier as well!
The familiar icon of chemistry, forced into awkwardness by the feature of showing "all the elements all the time", can not be readily replaced for learning and work, but the Alexander Arrangement, taught first, preserves the Table's validity, making sense of the need for breaks and gaps in the familiar form – flattened for convenience of printing and distribution – as printed projections of the World on maps are far handier as well!
|

