Before the recent 'rediscovery' of the three–dimensional periodic chart, the conventional flat periodic table served generations of students and chemists well, providing the basis, not only for the introduction to understanding chemistry, but a guide throughout the career of chemists, biologists, and teachers.
The 3D PERIODIC TABLE
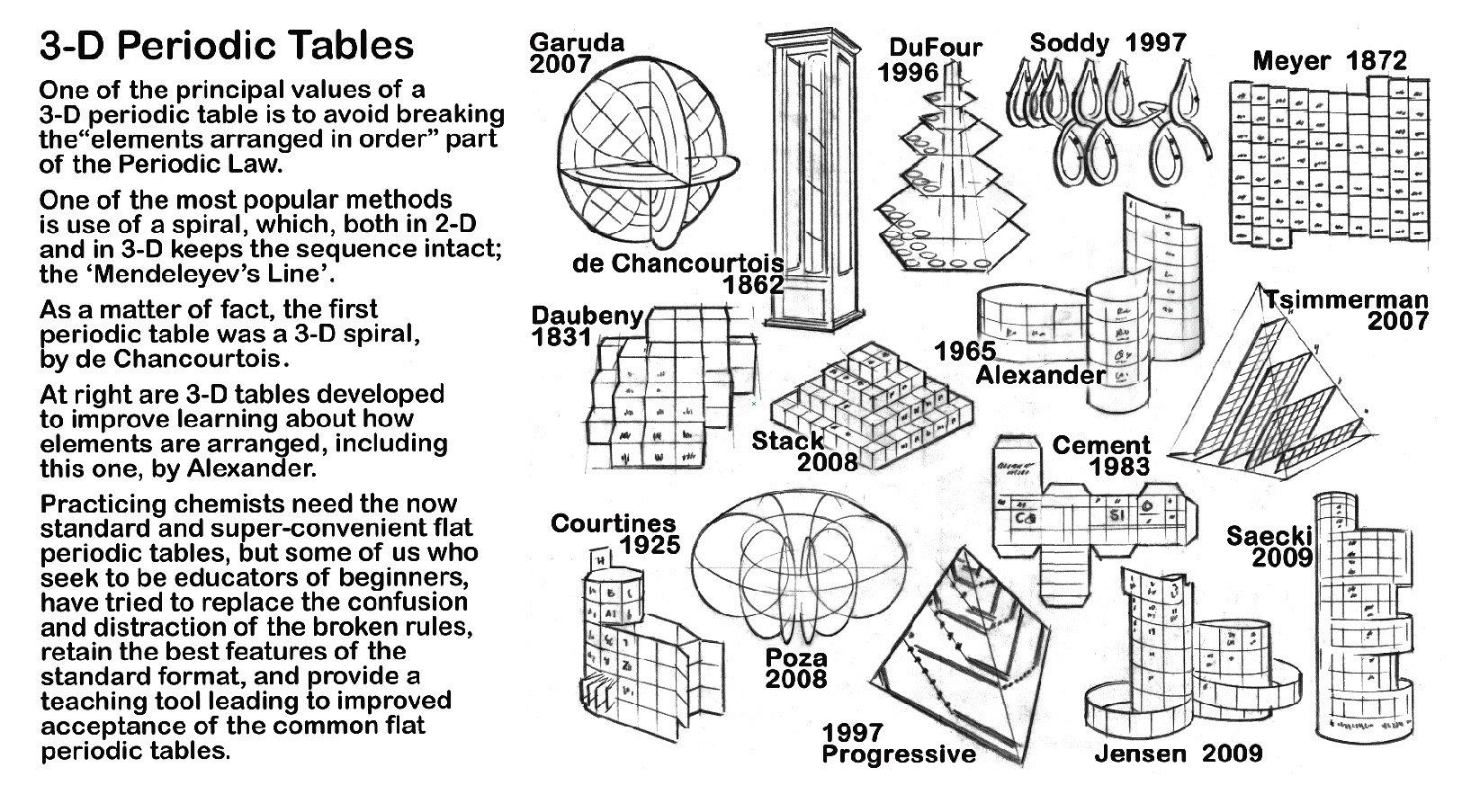
In spite of it's usefulness, however, difficulties in learning and using it have led to many efforts to improve the table. The elements have been placed in circles & spirals, duplicated elements, step–pyramids, trees, and target shapes with extensions. They have been formed into three–dimensional cubes, pyramids, stacks, even teardrops. Several have excelled the standard periodic table for accuracy and suitability in highly technical ways.
Falling partially within the spiral category, but primarily a helix, a 3D arrangement was designed by Roy Alexander in 1965, patented, and models first published in the mid–1990s. Three-dimensional creations by Roy Alexander, M.Courtines, and George Gamov, are the first modern element arrangement models to retain all the positive features of the flat table while removing ALL discontinuities and displacements in the order of the elements. The most recent Alexander Arrangement publication, called the Forever, showcases the marvelous element photographs of Theodore Gray on a model (from a papercraft kit) that also abbreviates the element list to those that exist NOW.




The 3D form, seen in a series of Alexander DeskTopper photos above, clearly defines, especially for beginning students, the new conclusions regarding the multi-positional aspects of hydrogen, the unity of the main group element's octet, and the separate nature of the d- and f-blocks.
These modern element arrangements are called the Alexander Arrangement of Elements, and discussion is under way about whether the form is that of the actual CHEMICAL ELEMENT SYSTEM, from which the row & column tables are projected.
A brief look at the dimensionality of periodic table design
© 2016 AlexanderDESIGN
4851 N. Washtenaw Ave., Chicago, IL 60645 773.271.0318
last update 5/24/16